Lesson 2: Origin and Occurrence of Hydrocarbons
2.0: Lesson Overview
Overview
Petroleum and natural gas are found within the pore-spaces of porous rock which forms the crude oil and gas reservoirs. Typically, the oil and gas are found with saline water which may be mobile or immobile. This is in contrast to the popular belief that crude oil is found as “oceans of oil” in vast underground caverns. The pore-space in porous rock is identical to the pore-space found within the sand grains of beach sand. If you were to dig a hole in the sand at the beach and fill it with ocean water, then that water would eventually drain out of the bottom of the hole and migrate through the sand back to the ocean. Oil and gas storage and transport are identical to the storage and transport of ocean water in beach sand. In fact, the physical laws that govern the storage and transport of fluids in porous rock and unconsolidated sand are identical.
Learning Objectives
By the end of this lesson, you should be able to:
- illustrate the types of geologic structures where crude oil and natural gas accumulate and/or exit;
- list the five elements that are required to form a hydrocarbon reservoir;
- describe the rock characteristics that are typically associated with hydrocarbons;
- explain the process by which hydrocarbons pass from source rock to reservoir trap;
- describe the geologic properties that make up a reservoir rock and what properties determine the commercial quality of the reservoir rock;
- identify the four hydrocarbon types that make up crude oil and natural gas;
- apply the Gibbs Phase Rule and explain how it governs the phase behavior of complex hydrocarbon mixtures;
- explain how the local hydrostatic gradient and thermal gradient determine the pressure and temperature of a hydrocarbon reservoir at discovery; and
- summarize how the location of the original pressure and temperature of the reservoir within the phase envelope of the phase diagram determines the type of crude oil or natural gas system that will be produced from the reservoir.
Lesson 2 Checklist
| To Read | Lesson 2: Origin and Occurrence of Hydrocarbons | Click the Basic Earth Geology link below to continue reading the Lesson 2 material |
|---|---|---|
| To Do | Submit the Lesson 2 quiz | Located in the Lesson 2 module in Canvas |
Please refer to the Calendar in Canvas for specific time frames and due dates.
Questions?
If you have questions, please feel free to post them to the Course Q&A Discussion Board in Canvas. While you are there, feel free to post your own responses if you, too, are able to help a classmate.
2.1: Basic Earth Geology
Since the occurrence of crude oil and natural gas are intrinsically associated with reservoir rocks, we must start our discussion of the origin and occurrence of natural hydrocarbons with petroleum geology. Geology is the study of all processes that affect the earth. The earth processes studied by geologists occur at many scales from those at the planetary scale (plate tectonics; interactions between the earth’s core, mantle, and crust; etc.) to those at the grain and pore scale (formation of the cementation that binds sand grains, swelling of clays in pores, etc.). Figure 2.01 shows a schematic diagram (not to scale) of the Earth’s Structure.

Through radiometric dating, the earth is believed to be approximately 4.54 billion years old. As shown in Figure 2.01, it is composed of an inner and outer core, the mantle, and the crust. The earth’s crust is the thin, solid, outer shell of the planet that we live on and interact with on a daily basis. The earth’s crust accounts for approximately 1% of the total volume of the planet.
The solid crust is the upper layer of the lithosphere (the lithosphere is composed of rigid crust and upper portions of the semi-elastic mantle). The earth’s crust can be further divided into the oceanic crust and the continental crust. The oceanic crust is between 3 miles (5 km) and 6 miles (10 km) thick and is overlain by approximately 3 miles of seawater; while the continental crust is between 20 miles (30 km) and 30 miles (50 km) thick. Underlying the oceanic crust and the continental crust is a layer of basalt. Because both the oceanic crust and the continental crust are less dense than the mantle, they both “float” on the mantle.
This floating of the crust on the mantle gives rise to the widely accepted theory of Plate Tectonics. Plate Tectonics is the theory that describes the motion of the continents over the geologic time scale. In this theory, the lithosphere is broken into several major tectonic plates and many smaller plates. The major plates are associated with one of the continents. Slow moving convection currents in the upper mantle are the primary driving mechanisms of plate tectonics; while other mechanisms related to gravity are considered to be secondary driving mechanisms. This is illustrated in Figure 2.02.
In this figure, four tectonic plates are shown along with the convection currents that are driving their drift. The direction of the tectonic drift is shown by the arrows. The separation of tectonic plates is referred to as rifting and is caused by the spreading of two or more convection currents. The mid-Atlantic Ridge is an example of rifting zone. Subduction of tectonic plates, where one plate slides below another plate, results when two or more tectonic plates traveling towards each other collide. The resulting zone is called a subduction zone. A third type of plate boundary, where one plate grinds past another plate, is called a transformational boundary. An example of a transformational boundary is the San Andreas Fault where the Pacific Tectonic Plate is moving across the North American Tectonic Plate.
Due to the friction and built-up stresses in subduction zones and transformational boundaries, these plate boundaries are often associated with volcanism and earthquakes. In fact, the “Ring of Fire” which surrounds the Pacific rim and the North American west coast is caused by the movement of tectonic plates interacting with the Pacific Tectonic Plate.
Rift zones and subduction zones are the only locations where mass transfer from the crust to the mantle (and vice versa) can occur. At a rift zone, fresh rock materials are expelled from the mantle; while at a subduction zone, weathered rock material is returned to the mantle. As we will see, this forms an integral part of the Rock Cycle.
2.2: Rock Types and the Rock Cycle
There are three primary rock types present on the earth: igneous, sedimentary, and metamorphic rocks.
Igneous rocks are rocks formed by the cooling and solidification of molten rock material. As such, igneous rocks are associated with volcanology. There are two types of igneous rocks, intrusive igneous rocks and extrusive igneous rocks. Intrusive igneous rocks crystalize below the earth’s surface, allowing for slow cooling and the development of large crystal structures, while extrusive igneous rocks crystalize on the surface, resulting in rapid cooling and the development of small crystal structures. Examples of intrusive igneous rocks include diorite, granite, and peridotite; while examples of extrusive igneous rocks include basalt, pumice, and tuff.
Sedimentary rocks are rocks formed by the accumulation of sediments. There are three types of sedimentary rocks: clastic sedimentary rocks, chemical sedimentary rocks, and organic sedimentary rocks. Clastic sedimentary rocks are rocks formed by the accumulation of debris that has been mechanically broken by earth processes such as weathering and erosion. Examples of clastic sedimentary rocks include sandstones, siltstones, and shales. Chemical sedimentary rock are rocks that are formed by the precipitation of dissolved materials from high mineral content brines. Examples of chemical sedimentary rocks include rock salt, chert, some limestones, and some dolomites. Finally, organic sedimentary rocks are formed from the accumulation of organic materials (dead plants or animals). Examples of organic sedimentary rocks include coal, some limestones, and some dolomites.
Metamorphic rocks are rocks that started as some other rock type (igneous or sedimentary) but have been substantially altered from their original form by excessive heat, pressure, or exposure to high mineral content brines (or combinations of these three mechanisms). Examples of metamorphic rocks include marble, gneiss, and schist.
As stated in the introduction section of this lesson, crude oil and natural gas reside in the pore-spaces between the grains that make up the rock fabric of the reservoir rock. Consequently, the overwhelming majority of hydrocarbon reservoirs occur in sedimentary rocks where the pore-spaces in the accumulated sediments are sufficiently large to allow for the storage of crude oil and natural gas. While the pore-spaces in metamorphic and igneous rocks are too small to allow for the entry of hydrocarbon molecules, if these rock formations are mechanically fractured by tectonic forces, then the space created by the natural fractures can allow for hydrocarbon storage, and fractured reservoirs in metamorphic and igneous rocks are possible.
It should be emphasized that the formation of rock is not a static process, but is more of a dynamic, cyclical process. In other words: when a rock is formed, it is continually acted upon by earth processes and is subject to change over the geologic time-scale. This process is referred to as the Rock Cycle and is illustrated in Figure 2.03.
2.3: Basic Petroleum Geology
The study of the geological processes that create crude oil and natural gas reservoirs is referred to as Petroleum Geology. In this discussion, we will also need to include brief discussions of related, specialized areas of geologic study including Stratigraphy and Structural Geology. Stratigraphy is the study of the layers (or strata) within rock formations; while Structural Geology is the study of the deformation of rock under tectonic forces. You are probably most familiar with the concepts of stratigraphy and structural geology from road-cuts seen along highways. These road-cuts often show cross-sectional outcrops of layered, deformed rock formations along the road-side. The layering you see is the local geologic stratigraphy; while the deformations you see form the local geologic structure of the rock formations.
Figure 2.04 shows a road-cut along Route 322 approximately 39 miles south of State College, PA. In this photograph, the layering (stratigraphy) and deformation (structure) of the Keefer Formation (Silurian Age) are clearly evident. The “inverted bowl-shaped” feature in this photo is an anticlinal structure, or simply an anticline. Anticlines statistically form the most common crude oil and natural gas reservoirs. We will discuss this in more detail when we discuss Reservoir Traps later in this lesson.
As we have already discussed, hydrocarbon reservoirs are typically associated with sedimentary rock formations. Over geologic time, weathered and eroded rock materials are carried downstream from elevated regions to lower regions in rivers and streams to oceans, seas, or lakes. At the point in the rivers, oceans, seas, or lakes where the energy in the water can no longer suspend or transport the rock material, it begins to settle in the water and is deposited onto the bottom of the water body. This is called the depositional process. During the depositional process, the environment can change over the geologic time scale: mountain tops and hilltops erode, sea-levels rise or lower, etc. With these changes, the types of sediments that are deposited change and the locations where they are deposited also change. Two commonalities that exist during the depositional process, even over the geologic time scale, are (1) the sediments and, consequently, the sedentary rocks that are lower in the stratigraphic column were deposited earlier than sediments higher in the stratigraphic column and (2) the layers of sediment are initially deposited horizontally. From this discussion, we can see that sediments lower in the stratigraphic column must be older than those higher in the stratigraphic column.
Note, a stratigraphic column is a written, pictorial, or schematic record of the local rock strata showing the order of the rock deposition (older rocks at the bottom and younger rocks at the top), along with notes concerning the age of the rocks, occurrence of any fossils, any rocks which may be absent from the local geological record due to erosion, or any notes of use to a geologist. One example of a stratigraphic column that you may be familiar with is The Obelisk on the University Park Campus of Penn State. The Obelisk is a physical column constructed of quarried building materials from Pennsylvania with the older rocks placed at the base of the column and the younger rocks placed at the top of the column. Consequently, it forms a true stratigraphic column of the building stones of Pennsylvania. A photograph of The Obelisk is shown in Figure 2.05.
From this discussion, we can also see that in their original, natural state, sedimentary rocks are horizontal. Any deformation (folding, uplifting, tilting, faulting, etc.) in sedimentary rock formations is caused by tectonic forces acting on the rock after deposition and solidification.
There are five geological requirements for the formation of a conventional hydrocarbon reservoir:
- Source Rock
- Migration Path
- Cap Rock
- Reservoir Rock
- Trap
These geological requirements are illustrated in Figure 2.06. It needs to be emphasized that although this figure consists of solid colored bands, these bands represent either porous or non-porous rocks. In the following discussions, all of the action concerning this figure is occurring in the pore-spaces of porous rock.
In this figure, the Source Rock is represented by the light grey layer. This is the rock in which the original organic material is converted into hydrocarbons. We will discuss this process later in this lesson. As we can see from this figure, the hydrocarbons do not necessarily originate in the hydrocarbon reservoir itself but are generated away from the reservoir in rocks that are conducive to hydrocarbon generation. As we will discuss later, these source rocks are typically organic-rich shales, siltstones, or coals.
Since the hydrocarbons are generated away from the reservoir, there must be a pathway for the hydrocarbons to migrate from the source to the reservoir. This pathway is the Migration Path in the hydrocarbon system. In the example shown in Figure 2.06, the migration path is a water-filled rock layer (aquifer) that is in communication with both the source rock the hydrocarbon bearing reservoir. This aquifer is shown as the blue layer in Figure 2.06.
In Figure 2.06, the solid green arrows represent the primary migration of the hydrocarbons, while the dashed green arrows represent the secondary migration of the hydrocarbons. Primary migration refers to the initial expulsion of the hydrocarbons from the source rock, while secondary migration refers to the remainder of migration to the reservoir.

As shown in this figure, all fluid migration is upward. This is because the main driving mechanism in hydrocarbon migration is buoyancy, which occurs because the oil and gas are less dense (lighter) than the resident water. In order to prevent this buoyant flow from occurring all of the way to the surface, a vertical flow barrier, or Cap Rock, is required along the migration path and at the reservoir itself. A cap rock is simply an overlying rock layer that is impermeable to flow. Permeability is a property of the rock that is a measure of the ease in which fluids can flow through a porous medium (in our case, a rock formation). In Figure 2.06, the cap rock is depicted by the black layer.
The fourth requirement for a hydrocarbon accumulation is the presence of a Reservoir Rock. In the example shown in Figure 2.06, the reservoir rock is the same rock formation as the migration path. As stated earlier, the most common reservoir rocks are sedimentary rocks; however, naturally fractured igneous and metamorphic rocks can also form hydrocarbon reservoirs. The two requirements for a commercial crude oil or natural gas reservoir are high porosity and high permeability. As discussed earlier, oil and natural gas exist in the pore-space between the grains of the sedimentary rocks. The porosity of a rock is defined as the fraction of the rock’s bulk (total) volume occupied by the pores. For example, a cubic foot (1 ft3) of rock with 15 percent porosity will contain 0.15 ft3 of pore space. Consequently, a rock formation with a higher porosity implies greater storage capacity than a rock formation with a lower porosity. This, in turn, results in the possibility of greater quantities of oil and gas stored in the more porous rock.
As discussed earlier, permeability is defined as the ease in which fluids flow through porous media. A high permeability formation implies greater oil and gas production rates and more economically attractive production wells.
The last component of a hydrocarbon system is the Trap. A trap or trapping mechanism is a change in the stratigraphy or a structural deformation that is capable of stopping the migration process and keeping the oil and gas in place over geologic time. In the example in Figure 2.06, the trap is an anticline like that shown in the photograph in Figure 2.04.
Statistically, anticlines are the most common traps; however, they are not the only types of hydrocarbon traps. Crude oil and natural gas traps can be categorized as Stratigraphic Traps or Structural Traps. As the names imply, stratigraphic traps are related to the layering of the rock strata; while structural traps are related to the structural deformations of the rock formations.
Examples of stratigraphic hydrocarbon traps are shown in Figure 2.07. Figure 2.07 shows (a) a “pinch-out” trap and (b) an unconformity trap. In these figures, the yellow layers represent reservoir quality rock (Reservoir Rock); while the green areas represent an oil accumulation in the reservoir trap.

A pinchout trap, Figure 2.07a, is formed due to the relative rise and fall of a water body in relation to the local land mass. (Note: a relative rise or fall may be due to either a true rise or fall of the sea level, or a subsidence or uplift of the land mass.) In this figure, the crude oil reservoir is represented by the green area encased in the black, impermeable layer. As mentioned earlier in this lesson, the size and weight of the sediments that form sedimentary rocks are deposited in water are dependent on the energy of the water suspending the particles. Heavier, coarser materials can only be suspended in high energy environments near the shore where wave and tidal action keep these materials mixed with the water. These materials are deposited as sediments at the point in the depositional environment where the wave and tidal energy can no longer support them. On the other hand, lighter, finer sediments can be suspended in low energy environments away from the shore in more calm waters and, consequently, are deposited further from the shoreline. As the relative position of the sea level changes over the geologic time scale, the positions of the coarser and finer sediments change. The coarser materials with the larger pore spaces between the sediment grains often make good quality rock, while the finer materials, particularly clays and silts, with smaller pore spaces make good impermeable seals (cap rocks). Under the proper sequence of events, as the geographical locations of the sediments change over the geologic time scale, the changes in the size of the sediments can create pinchout, or stratigraphic trap.
Figure 2.07b shows an unconformity trap. A geological unconformity is a boundary between two rock sequences of different ages that are missing some transitory rock sequences of intermediate ages - in other words, some rock strata are missing from the local geological record. This gap in the geological record is caused by a past erosional period.
The depositional history of an unconformity trap is slightly more complicated than that of a pinchout trap. In this trap system, an early depositional period caused sedimentary rocks to be deposited horizontally. These rocks were then tilted due to local tectonic forces. These form the lower tilted layers in Figure 2.07b. The higher, uplifted portions of the tilted layers were then eroded over geological time. The orange wavy line represents an unconformity surface where rocks are missing from the geological record. Geologists recognize these unconformity surfaces when they see rocks of one age sitting on top of rocks of a different age with no intermediary age rocks between them; while paleontologists recognize unconformities by rocks containing fossils from one age sitting directly on top of rocks containing fossils from a different, much older age. After subsidence, a second depositional period occurs where additional sedimentary rocks are again deposited horizontally over the erosional surface. In order for an unconformity trap to develop, the first layer deposited during the second depositional period must be an impermeable rock which can act as the cap rock for the reservoir. After the second depositional period, the trap can be charged with crude oil to form the oil reservoir - the green area of the figure. (Note that the horizontal yellow layer representing good quality reservoir rock above the unconformity surface, does not contain crude oil or natural gas. This is because it is missing a trapping mechanism at this location.)
These histories illustrate the critical nature of the timing in the development of hydrocarbon reservoirs. For a hydrocarbon reservoir to develop, all five elements of the reservoir system must be in place prior to the formation and migration of the hydrocarbons. If one element is missing from the system, then a crude oil or natural gas reservoir cannot develop.
The second major category of hydrocarbon reservoirs is structural traps. Structural traps, illustrated in Figure 2.08, are related to mechanical deformations and failures of the reservoir rock. Figure 2.08a shows an anticlinal structure, or anticline. As stated earlier, the anticline is statistically the most common reservoir trap. The reason for this is very simple. An anticlinal trap simply requires that the reservoir rock and cap rock be folded in a manner that can trap the lighter hydrocarbons. Figure 2.04 shows the Arch Rock anticline near State College, PA.
Figure 2.08b shows a fault trap. A fault trap is created when a rock formation undergoes mechanical failure during an earthquake and the resulting fault causes a porous and permeable rock formation (reservoir rock) to be positioned adjacent to an impermeable rock formation. Thus, closure to the trap is formed by the cross-fault impermeable layer.
Figure 2.08c shows traps along a salt dome. These traps are common in areas where salt formations exist, such as in the Gulf of Mexico. Since large volumes of salt behave in a plastic manner over geologic time, the weight of the overburden creates great pressures within the salt causing it to flow through any local weak spots in the overburden. These salt intrusions can create hydrocarbon traps above and to the flanks of the intrusion as depicted in Figure 2.08c.

2.4: Origins of Oil and Natural Gas
To this point, we have discussed the elements required for the formation of a hydrocarbon reservoir. We have also discussed the timing and histories of the formation of the reservoir traps. We have also mentioned that all of the elements of the hydrocarbon reservoir must be in place prior to the formation and migration of the hydrocarbons. We are now able to discuss the actual formation of the hydrocarbons themselves.
Petroleum and natural gas are naturally occurring substances composed of hydrocarbon molecules (molecules made up of hydrogen and carbon atoms) and, possibly, non-organic contaminants, such as CO2, H2S, N2, and O2.
The most widely accepted theory of the origin of petroleum and natural gas is the Organic Origin Theory which states that these fluids are generated from the decay of prehistoric plants and animals under the influence of the excessive pressures and temperatures that exist in the earth’s subsurface. Research indicates that petroleum and natural gas originated from zooplankton (single-celled plants found drifting freely in fresh or brackish water) and algae. During their lifetimes, these organisms create energy from photosynthesis to carry out their life processes, and it is this energy that we use today (thus the term, “fossil fuels”). Contrary to the popular myth, petroleum and natural gas do not originate from decomposed dinosaurs.
Over the geologic time scale, these free-floating zooplankton and algae die, settle to the ocean-, sea-, lake‑, river‑, or swamp-bottom, and accumulate as sediment. This process occurs simultaneously with the geological processes (erosion, transportation, and deposition) acting on the sand, silt, and clay sediments that form sedimentary rocks. As the organic material and rock materials accumulate on the sea bottom, the materials at the top of the accumulation exert increased weight (pressure) on the materials at the bottom of the accumulation. As this depositional process continues and the accumulation becomes thicker, the impact of the earth’s geothermal gradient also begins to act on the organic materials. At elevated pressures and temperatures, the decaying organic materials are transformed into a dark waxy material called kerogen. Kerogen is an intermediary stage in the development of petroleum. The process of converting the original organic material into kerogen is called the Catagenesis Process. The formation of kerogen requires that the rock containing the original organic material be impermeable. This requirement is essential because as kerogens are formed, they must remain trapped within the pore-spaces of the rock and cannot be allowed to escape to other rock formations or to the environment. As we have already discussed, the rock formation in which the original organic materials are converted to kerogen, and eventually petroleum, are called source rocks for the hydrocarbons. Typically, fine-grained, clay-rich sedimentary rocks, such as shales, are the most common source rocks for hydrocarbon basins because they satisfy two of the requirements for kerogen generation; they (1) are sedimentary rocks and (2) contain adequately low permeabilities.
During the catagenesis process, the kerogen is “cooked” in the high-temperature environment, and the long-chain kerogen molecules are “cracked” into shorter-chained hydrocarbon molecules. It may take several million years for the deposition process to bury the original organic materials and the source rock to achieve the appropriate temperatures (> 250 - 300 ºF) for kerogen generation and another several million years to generate commercial quantities of oil and gas. There is a specific temperature range, referred to as the “oil window,” in which oil is formed. At temperatures below the oil window, the catagenesis process cannot occur; while at higher temperatures the “cooking” and “cracking” processes are stronger and very short-chain natural gas molecules are generated (thermogenic gas).
A second, less accepted theory for the origin of hydrocarbons is the Inorganic or Abiogenic Theory. In this theory, hydrogen and carbon from inorganic sources are fused at the elevated pressures and temperatures in the earth’s subsurface and are converted to hydrocarbons. While the presence of inorganically sourced hydrocarbons cannot be discounted, there is abundant evidence that the vast majority of crude oil and natural gas have an organic origin.
2.5: Hydrocarbon Types
Crude oil is a complex mixture of several types of hydrocarbon molecules along with inorganic impurities. These hydrocarbon molecules are:
- Paraffinic (or alkane series) hydrocarbons
- Naphthenic (or saturated cyclic) hydrocarbons
- Aromatic (or cyclic) hydrocarbons
- Asphaltene hydrocarbons
Sometimes we refer to a particular crude oil as a paraffinic crude oil or as an aromatic crude oil. While there may be several thousand different hydrocarbon molecules in a given crude oil, these descriptions simply refer to the dominant hydrocarbon type in the mixture. All naturally occurring crude oils will typically contain molecules of each type.
Paraffinic, or alkane series, hydrocarbons are shown in Table 2.01. These hydrocarbons are typically studied in introductory courses in Organic Chemistry. Alkane series hydrocarbons are composed of only hydrogen and carbon atoms attached with single bonds. They are characterized by the following formula for the number of hydrocarbon atoms present in the molecule:
In this formula, NH is the number of hydrogen atoms, while NC is the number of carbon atoms in the hydrocarbon molecule.
Normal alkanes are the chain molecules depicted the first four examples in Table 2.01. Once the number of carbon atoms reaches four (butanes) different permutations of an alkane molecule can exist that still honor Equation 2.01 but do not form chain molecules. These permutations are called isomers with the number of possible permutations (and number of isomers) increasing as the number of carbon atoms, NC, increases. An example of the difference between a normal and isomeric alkane is shown in Table 2.01 for n-butane and i-butane. Due to the slight discrepancies in the molecular structures, normal alkanes and isomeric alkanes have slightly different physical and chemical properties, such as, boiling points, melting points, etc.
| Compound | Formula | 2D Representation | 3D Representation |
|---|---|---|---|
| Methane | CH4 |
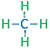 |
 |
| Ethane | C2H6 |
 |
 |
| Propane | C3H8 |
 |
 |
| N-Butane Normal Butane |
C4H10 |
 |
 |
| I-Butane Isomeric Butane |
C4H10 |
 |
 |
As the number of carbon atoms increases, the molecular weight of the molecule increases. The lower molecular weight alkanes, methane and ethane, are the most common hydrocarbon components in natural gas; however, intermediate molecular weight alkanes up to the butanes may also be present in natural gases. In addition to these hydrocarbon components, some inorganic impurities, such as CO2, H2S, N2, and O2 may also be present in natural gases.
Crude oils from Pennsylvania are generally classified as Paraffinic Crude Oils (“Pennsylvania Grade Crude Oil” was at one time a phrase used to describe high quality crude oils.). As a solid, the alkanes form a waxy substance, paraffin, which is the main component of such products as paraffin candles.
The second category of hydrocarbon molecules found in crude oils are naphthenic (or saturated cyclic) hydrocarbons. Like the alkanes, these hydrocarbons are composed of hydrogen and carbon atoms attached with single bonds; however, they differ from the alkanes in that they do not form chain structures but form cyclic (ring) structures. Examples of naphthenic hydrocarbons are shown in Table 2.02.
| Compound | Formula | 2D Representation |
|---|---|---|
| Cyclopropane | C3H6 |
 |
| Cyclobutane | C4H8 |
 |
| Cyclopentane | C5H10 |
 |
The third category of hydrocarbon molecules found in crude oils are the aromatic (or cyclic) hydrocarbons. These hydrocarbons are composed of hydrogen and carbon atoms that form cyclic structures but contain dual bonds between alternating carbon atoms. The simplest aromatic hydrocarbon is benzene with a single ring structure; while more complex aromatic hydrocarbons are typically formed with multi-ring structures. As the name implies, aromatic hydrocarbons, such as benzene, are associated with a sweet smell. Examples of aromatic hydrocarbons are shown in Table 2.03.
The last category of hydrocarbon molecules found in crude oils are the asphaltene hydrocarbons. These hydrocarbons are large, high molecular weight molecules which may also contain some atoms other than hydrogen and carbon atoms, such as, sulfur, oxygen, or nitrogen atoms. Asphaltenes typically result in the residue from the refining process and are the principal components of asphalt (road paving), tar, and bitumen products.
The mixture of these hydrocarbon molecules can have several consequences on oil and gas production, transport, and refining. These include:
- The mixture of hydrocarbon molecules gives the bulk fluids (oil and gas phases) the properties which dictate the ease or difficulty in which they flow through the reservoir towards production wells and through production equipment
- The nature of any solids which may precipitate from the bulk fluids onto rock grains, wellbore, or surface equipment during production causing operational problems requiring different solutions:
- Paraffinic crude oils may deposit waxy solids onto the reservoir rock, well completion, or production tubing causing restrictions to flow requiring a remediation treatment, such as hot kerosene injection into the well.
- Asphaltic crude oils may deposit hydrocarbon resins onto the reservoir rock, well completion, or production tubing causing restrictions to flow requiring a remediation treatment, such as solvent injection into the well.
- Local field treatment of the crude oil to upgrade it to the required specifications for the pipelines and refineries where the crude oil will eventually flow
- The products that can be refined from the crude oils once they reach the refineries: lubricating oils, heating oils, car or aviation fuels, asphalt and tar products, etc.
| Compound | Formula | 2D Representation |
|---|---|---|
| Benzene | C6H6 |
 |
| Napthalene | C10H8 |
 |
The definitive methods for determining the different components in crude oil are with laboratory measurements, such as gas chromatography. Often, however, less rigorous methods may be useful for quick, on-site evaluations or for numerical (computer) calculations where the crude oil type is characterized with a single parameter. In the oil and gas industry, one common measure of the dominant character of the crude oil (paraffinic, naphthenic, and aromatic) is the Watson Characterization Factor[1] (or a generalization suggested by Whitson[2]). The original form of the Watson Characterization Factor, KW, is:
Where:
KW is Watson Characterization Factor, oR1/3
Tb is the mean average boiling point of the mixture, oR
ϒo is the specific gravity of the mixture, dimensionless
This equation requires that the boiling point, Tb, for the mixture of interest is known. In many cases, Tb is unknown or difficult to measure. For these cases, Whitson correlated the Watson Characterization Factor with the more commonly known molecular weight:
Where:
KW is Watson Characterization Factor, oR1/3
Mo is the molecular weight of the mixture, lbm/lbm-mol
ϒo is the specific gravity of the mixture, dimensionless
The guidelines for the use of the Watson Characterization Factor are:
- KW ≥ 12.5 indicates a paraffinic crude oil
- 12.5 > KW ≥ 10 indicates a crude oil containing naphthenic or aromatic components
- KW < 10 indicates a highly aromatic crude oil
Table 2.04 shows the values of the Watson Characterization Factor for substances of known hydrocarbon type (paraffinic, naphthenic, and aromatic).
| Hydrocarbon Series |
Substance | Formula | Tb (oR) |
Mo (lbm/lbm-mol) |
Υo |
Kw(oR1/3) Eq. 2.02a |
Kw(oR1/3) Eq. 2.02b |
|---|---|---|---|---|---|---|---|
| Paraffin | n-Hexane | C6H4 | 615.4 | 86.178 | 0.6640 | 12.8 | 12.7 |
| 2-Methylpentane (A) | C6H14 | 600.1 | 86.178 | 0.6759 | 12.8 | 12.8 | |
| n-Heptane | C7H16 | 668.8 | 100.205 | 0.6882 | 12.7 | 12.6 | |
| Napthene | Cyclohexane | C6H12 | 637.0 | 84.162 | 0.7834 | 11.0 | 11.0 |
| Methylcyclohexane | C7H14 | 673.4 | 98.189 | 0.7740 | 11.3 | 11.4 | |
| Aromatic | Benzene | C6H6 | 635.8 | 78.114 | 0.8844 | 9.7 | 9.8 |
| Toluene | C7H8 | 690.8 | 92.141 | 0.8718 | 10.1 | 10.2 |
(A) One of the isomers is Hexane
2.6: Types of Hydrocarbon Reservoirs
To this point in the lesson, we have discussed the origins of the reservoir rocks and traps that formed over the geologic time scale which result in modern oil and gas reservoirs. We have also discussed the origins of the hydrocarbons that reside in pore spaces of these reservoirs. We can now discuss the types of reservoirs and reservoir systems that are currently of commercial interest to the oil and gas industry. To aid in this discussion, we must first introduce the concept of the Phase Envelope (or, Phase Diagram, or Pressure-Temperature Diagram, or P-T Diagram) for single-component (pure) and multi-component (mixture) systems.
2.7: The Gibbs Phase Rule
The Gibbs Phase Rule relates the degrees of freedom in a system to the number of components and number of phases in a system. The Gibbs Phase Rule is:
Where:
F the number of degrees of freedom in the system, integer
C is the number of components in the system, integer
P is the number of phases in the system, integer
The use of the Gibbs Phase Rule is best illustrated with examples; however, to do this we must first discuss some fundamental thermodynamic concepts. The phrase “degrees of freedom” refers to the maximum number of independent thermodynamic variables (pressure, temperature, and intensive variables) that you can vary simultaneously within a system at equilibrium before you are forced to specify one or more of the remaining variables (or disturb the equilibrium of the system). By “intensive variables”, we are referring to variables that are independent of the size of the system. For example, phase density in a system is an intensive variable because you can halve the size of the system, and the phase density will remain the same. On the other hand, the mass of a system is an extensive property because if you halve the size of the system, then you halve the mass in the system. A component refers an individual chemical element that exists in the system (in our case, a molecular species: methane, ethane, cyclopentane, benzene, CO2, H2O, etc.). Finally, a phase is a physical state of matter with homogenous (uniform) composition, physical properties, and chemical properties. In petroleum and natural gas engineering, we typically deal with systems containing four phases: a gaseous hydrocarbon phase (natural gas), a liquid hydrocarbon phase (crude oil), a liquid aqueous phase (water or brine), and a solid rock phase. There are times when we deal with systems containing more phases (such a solid hydrocarbon phase or multiple hydrocarbon liquid phases), but the circumstances when this occurs are beyond the scope of this class.
For a single-component system, we have C = 1, and Gibbs Phase Rule becomes:
If we were to plot the phase state (number and types of phases) on a pressure-temperature plot for this single-component system, then we would obtain a plot like that shown in Figure 2.09. This figure is also referred to as a Phase Diagram or a P-T Diagram. In this Phase Diagram, the grey region represents all of the pressure-temperature combinations that result in the solid form of our single component, the green region represents the pressure and temperature combinations that result in the liquid form of our single component, the red region represents the pressure and temperature combinations that result in the gaseous form of our single component, and the multi-colored region represents the super critical form of our single component. Also posted on this Phase Diagram are two points, the triple point and the critical point.
Getting back to the single component version of the Gibbs Phase Rule, Equation 2.04, we can start to clarify some of the concepts that we have already discussed with some examples. If we consider the single-phase regions in Figure 2.09 (P = 1 in Equation 2.04), then from Equation 2.04, we have 2 degrees of freedom (F = 2). This implies that we can independently vary Pressure and Temperature within the Phase Envelope (colored regions) and not change the phase state of the system. In other words, two degrees of freedom represent two-dimensional regions (areas) on the Phase Diagram in which a single phase exists.
Now, let’s consider the occurrence of two phases coexisting simultaneously (P = 2) in equilibrium. From Equation 2.04 with P = 2, we have one degree of freedom (F = 1). In Figure 2.09, we have two coexisting phases along the borders of the phase envelopes. For example, the line bordering the Solid and Liquid Phases represents the pressure-temperature conditions where the solid form of our single component system can coexist with the liquid form of our single component at equilibrium (think of polar caps sitting on water near the earth’s poles). For two phases to coexist in equilibrium, if we were to change one variable, say temperature, then we would be forced to change pressure in a manner that it remained on the border line between the two single-phase regions. In other words, one degree of freedom represents the one-dimensional, curvilinear lines which act as borders between the single-phase regions. If we change one variable on one of these border lines, then we are forced to change the other variable to remain on the border line.

Finally, we can consider the coexistence of three phases (P = 3 in Equation 2-4) in a system at equilibrium. From Equation 2.04 with P = 3, we have F = 0, or no degrees of freedom. This implies that at the point where three phases coexist (the Triple Point in Figure 2.09), we cannot change either pressure or temperature and retain a three-phase system in equilibrium of the single-component (pure) system. The Triple Point is a property of the component that we are considering. Thus, zero degrees of freedom refers to a 0-Dimensional point (the Triple Point)
In Figure 2.09, the Critical Point is also plotted. The Critical Point, defined by the critical pressure, Pc, and the critical temperature, Tc: (Pc, Tc) is the point in the system that defines the onset of the super critical state. Super critical fluids are fluids in which the gaseous phase becomes indistinguishable from the liquid phase (the phase densities become equal).
Consider a pressure-temperature pair in the single-phase gas region with a temperature somewhere between the temperature at the triple point and the critical temperature, Tc, such as Point A in Figure 2.10. If we were to increase the pressure of this single-phase gas (Path A-A’), then we would see a distinct and abrupt change in the phase state of the fluid as we crossed into the single-phase liquid region. This is because the densities of the gaseous phase and the liquid phase are different. Now, if we were to do the same experiment starting at a temperature greater the critical temperature, Tc, such as Point B, then as we increased the pressure on the single-phase gas (Path B-B’) and entered the Super Critical Fluid region, there would be no abrupt phase change.
We could also perform a third experiment where we started at the original point, Point A, and followed the pressure-temperature path Path A-B-B’-A’ into the single-phase liquid region to arrive at Point A’. In this last experiment, we would arrive at the same point, Point A’, as in the first experiment, but without any abrupt phase change. The system properties would change smoothly and continuously during the entire experiment.

For multi-component systems like real crude oil – natural gas systems, the Pressure-Temperature Diagram is much more complex. This is because a real crude oil – natural gas system may contain tens or hundreds of thousands of components. For Multi-Component Systems, the P-T Diagram looks like that in Figure 2.11. A Phase Diagram, such as that shown in this figure, typically is measured in the laboratory but can also be generated mathematically with sophisticated models, such as, Cubic Equation of State Equations (multi-component extensions to van der Waal’s Equation).

In this figure, the region between the green curve and the red curve is the two-phase envelope; while the region outside of the two-phase envelope is the single-phase region. Single-phase liquids exist to the left (lower temperatures) and above (higher pressures) the bubble-point locus (green curve); while single-phase gases exist to the right (higher temperatures) of the dew-point locus (red curve).
The green curve in this figure represents the Bubble-Point Locus of this multi-component system; the red curve represents the Dew Point Locus of the system, and the dashed lines in the two-phase region represents the quality lines of the system (lines of constant volume-fraction of the liquid phase). The bubble-point, defined by a bubble-point pressure, Pb and a bubble-point temperature, Tb, is the point on a pressure-temperature path (originating in the single-phase liquid region) where the path enters the two-phase region (crosses the green curve in Figure 2.11). The name “bubble-point” comes from the fact that this is the point where the first bubble of gas evolved from a liquid as it enters the two-phase region. For example, Point A in Figure 2.11 lies in the single-phase liquid region. As the pressure is reduced at a constant temperature (isothermal conditions), it follows the Path A-A’. The point where Path A-A’ enters the two-phase region (crosses the green, bubble-point locus) represents the bubble-point pressure for this temperature. This is the point where the first bubble of gas is formed in the system. The pressure reduction is continued until it terminates at Point A’. Point A’ lies on the 0.9 quality line, implying that at this point, the system is composed of two-phases with the liquid phase occupying 90 percent of the volume.
On the other hand, the dew-point, defined by the dew-point pressure and dew-point temperature is the point on a pressure-temperature path (originating in the single-phase gas region) enters the two-phase region. The name dew-point refers to the point where the first liquid drop condenses from the gaseous phase. For example, Point B in Figure 2.11 lies in the single-phase gas region and enters the two-phase region when the pressure is reduced under isothermal conditions (Path B-B’). The dew point pressure for this temperature then is the pressure where Path B-B’ crosses the red dew-point locus. If the isothermal pressure reduction is stopped at Point B', then the system at equilibrium will contain two phases with the liquid phase occupying 10 percent of the system volume.
Finally, the pressure, Pmax in Figure 2.11 is the cricondenbar (the maximum pressure in which two phases can coexist); while the temperature, Tmax is the cricondentherm (the maximum temperature in which two phases can coexist).
With these preliminary concepts, we can now continue with our discussion on the types of hydrocarbon reservoirs encountered in the oil and gas industry.
2.8: Undersaturated Black Oil (Low Shrinkage Oil) Reservoirs
Undersaturated black oils, sometimes referred to as “low shrinkage oils,” are single-phase liquid systems that reside in reservoirs with an original temperature that is significantly lower than critical temperature, TC. The phase diagram for an undersaturated black oil is shown in Figure 2.12.
In this figure, the reservoir temperature, TR, and reservoir pressure, pR, at the time of discovery are determined by the earth’s local temperature gradient and hydrostatic pressure gradient, respectively. The phase diagram for the crude oil is determined by the composition of the crude oil. As shown in this figure, the original reservoir temperature and pressure lie in the single-phase liquid region of the phase diagram at a temperature lower than the critical temperature.
The term “undersaturated” implies that the crude oil has the capacity to dissolve additional gaseous components and remain in the single-phase region. For example, if we change the composition of the system by adding more lower molecular weight components (e.g., methane and ethane), then the phase envelope would expand with the bubble-point locus beginning to shift towards the Point (TR, pR). In this example, we could continue to add gaseous components to the system until the bubble-point locus has shifted all of the way to the Point (TR, pR). At this point, with Point (TR, pR) lying on the bubble-point locus, we would call the fluid a bubble-point fluid. This is the point where the system would be unable to accept additional gaseous components and still remain a single-phase liquid, i.e., a single-phase crude oil. We will discuss the significance of the term “low shrinkage” when we discuss “high shrinkage” volatile oils.
The production of oil and gas is an isothermal process. We typically deplete the reservoir pressure by removing fluids from the system. Even in the cases where we inject fluids at different temperatures into the reservoir, such as with steam injection, the volume of fluids that we inject are so small compared to the size of the reservoir and the over- and under-burdens that, at most, we cause a local change in the temperature at the point of injection and have no significant impact on the temperature of the system. For isothermal processes, the pressure-temperature path of the fluids inside the reservoir is a vertical line at a fixed temperature. This is represented in Figure 2.12 (A) as the solid black arrow going from Point (TR, pR) to Point (TR, pA).

Figure 2.12 (B) shows the undersaturated black oil in an anticlinal trap at the time of discovery, that is, at the original reservoir temperature, TR, and pressure, pR. This figure depicts a single-phase liquid hydrocarbon (crude oil) system. As we produce reservoir fluids, single-phase oil (and the dissolved natural gas components) and possibly water, the pressure in the reservoir depletes. This is the solid arrow from Point (TR, pR) to Point (TR, pA). At some point in time, the reservoir pressure drops below the bubble-point pressure, and we enter the two-phase region in the phase diagram. At this point in time, a second, free gas phase evolves in the reservoir. This occurs when the path of the reservoir fluids crosses the green, bubble-point locus.
As we continue production, we now produce crude oil (and the gas dissolved in it), free natural gas, and, possibly, water. At some point during the depletion process, the natural pressure in the reservoir becomes so low that it can no longer overcome the weight of the fluids in the wells and lift these fluids to the surface at commercial rates. At that point in time, we may be able to install some form of artificial lift (e.g., pumps) to help the natural reservoir pressure lift the reservoir fluids, but that is only a temporary solution, and eventually we will be forced to abandon the reservoir. The pressure at which we abandon the reservoir is called the “abandonment pressure,” pA.
Figure 2.12 (C) shows the same reservoir at abandonment conditions after the free gas phase has developed. Since gas is lighter than oil, it will migrate to the top of the reservoir and accumulate there. When gas comes out of solution due to pressure depletion and accumulates at the top of a reservoir, we refer to the resulting accumulation as a “secondary gas cap.”
Please note that we may run into some situations in which the abandonment pressure occurs before we drop below the bubble-point pressure. In these cases, the path of the reservoir fluids would remain in the single-phase liquid region, and a free gas phase would never develop in the reservoir.
Also depicted in Figure 2.12 (A) is the dashed arrow from Point (TR, pR) to Point (TS, pS). This is the path that the reservoir fluids travel in the well and through the surface facilities. Point (TS, pS) refers to the separator conditions. The separators are field equipment, typically designed by facilities engineers, that are used to separate the produced fluids. Normally, produced oil has a higher commercial value to the oil and gas company than produced natural gas, so typically the facilities engineers design the separators to optimize the volume of liquid at the surface. In other words, the faculties engineers will design the separator conditions TS and pS to lie on the quality line of the phase diagram with the maximum feasible volume percent liquid.
2.9: Saturated Black Oil (Low Shrinkage Oil) Reservoirs
Saturated black oils are oils which are fully saturated with natural gas components (e.g., methane and ethane). This implies that the original reservoir temperature and pressure are in the two-phase region of the phase diagram at the time that the reservoir is discovered. This is shown in Figure 2.13.

Figure 2.13 (B) shows a saturated black oil in an anticlinal trap. In this figure, we can see that a gas cap overlies the oil at discovery. Since this gas accumulation was present at the original temperature and pressure conditions, we refer to this gas accumulation as a “primary gas cap.” Figure 2.13 (C) shows the state of the reservoir at abandonment conditions. As shown in this figure, at the lower pressure condition, the gas cap size has expanded.
2.10: Undersaturated Volatile Oil (High Shrinkage Oil) Reservoirs
Undersaturated volatile oil reservoirs are single-phase oil systems that reside in reservoirs with an original temperature relatively close to the critical point (relative compared to low shrinkage oils). As with the undersaturated black oils, the term undersaturated implies that the original reservoir temperature and pressure lies in the single-phase liquid region of the phase diagram. Such a system is shown in Figure 2.14.

We are now in a position to discuss the terms “low shrinkage oil” and “high shrinkage oil” as they pertain to the oil and gas industry. The blue bars on Figure 2.14 (A) represent equal pressure drops below the bubble-point pressure (these bars are of equal length). We can see from this figure that due to the convergent nature of the quality lines (dashed lines) near the critical point, equal pressure drops result in different volume percentages of the liquid phase in the two-phase region. The pressure drop represented by the blue bar further from the critical point results in a two-phase system with 90 percent of the volume occupied by the liquid phase; while the blue bar closer to the critical point results in a two-phase system with 80 percent of the volume occupied by the liquid phase. Thus, the volume of the liquid phase shrinks more in a system that is closer to the critical point for similar pressure drops. Since the liquid phase occupies less volume for a volatile, high shrinkage oil for comparable pressure drops, at abandonment the secondary gas cap will occupy a larger volume.
The definition of a “volatile fluid” is a fluid that easily evaporates or vaporizes. This definition gives rise to the name “volatile oils.” These systems develop larger gas phase volumes for comparable pressure drops when entering the two-phase region of the phase envelope.
2.11: Gas Condensate Reservoirs
Gas condensate reservoirs are gas systems that reside in reservoirs with the original temperatures lying between the critical temperature, TC, and the cricondentherm (Tmax in Figure 2.11). If the original reservoir pressure is greater than the dew-point pressure, then a single-phase gas system will occur in the reservoir as in Figure 2.15.
In this figure, the reservoir is initially a single-phase gas, but as we deplete the pressure due to gas production the pressure-temperature path of the system enters the two-phase region of the phase envelope and liquid hydrocarbons condense from the gas in the reservoir (thus the name gas condensate reservoir.)

We are actually very familiar with this process in our everyday lives. Think of a four-component mixture of oxygen, carbon-dioxide, nitrogen, and water vapor (a mixture of air) and its phase envelope. If we started at a point in the single-phase gas region with a pressure below the cricondenbar (pmax in Figure 2.11) and reduced the temperature under constant pressure conditions (isobaric conditions), then we would cross the dew point locus of the phase envelope. This would be the pressure and temperature where we would see the first dew forming on plants and the first water condensation forming on glass and metal. If we were to continue reducing the temperature, then the percent volume of liquid (water) would increase and it would begin to rain. What is happening in this simple example is that the heaviest component in our system (water vapor) is condensing out of the gas phase (air mixture) and forming a second phase (liquid water) in the two-phase region.
This is essentially what is occurring in the reservoir of a gas condensate system but under isothermal conditions. As we pass through the dew point pressure, the heaviest hydrocarbon components in the system begin to drop out and form a second, liquid hydrocarbon phase in the two-phase region of the phase envelope inside the reservoir.
There is one interesting characteristic of gas condensate reservoirs that is worth further discussing and that is the retrograde behavior of these systems. This is illustrated in Figure 2.16. In this figure, if we were to follow the isothermal Path A-A’-A’’, then we would go below through the dew-point pressure, increase the volume percentage of the liquid hydrocarbon phase until it reached a maximum at Point A’ with further reductions in pressure resulting in a lower volume percentage of the liquid hydrocarbon phase. We could also continue the isothermal pressure reduction, reenter the single-phase gas region, and stop at Point A’’.
We can see from Figure 2.09 and Figure 2.10 that for a pure (single-component) system, the liquid phase occurs at higher pressures than the gas phase. Thus, if we were to start in the single-phase liquid region of a pure system, we would need to reduce the pressure isothermally to create a gas phase.
The analogy for our multi-component system is that if we start at the point of maximum liquid volume (Point A’ in Figure 2.16) and reduced the pressure isothermally, then we would get the conventional behavior for a pure system along Path A’-A’’. Conversely, if we were to start at Point A’ and increased the pressure isothermally to Point A in the single-phase gas region, then we would get the behavior opposite of that for a pure system along Path A’-A. This behavior, opposite to a pure system, is referred to as retrograde behavior. This behavior occurs in the green shaded region in Figure 2.16. This region, formed by connecting all of the points of maximum temperature on the quality lines, is referred to as the retrograde region of the fluid.

Sometimes we see or hear the terms “Retrograde Condensate Reservoir” or “Retrograde Condensate System.” This terminology is used to describe reservoirs in which the isothermal reservoir pressure-temperature path traverses the retrograde region of the system’s phase envelope.
2.12: Wet Gas Reservoirs
Wet gas reservoirs are gaseous hydrocarbon systems that reside in reservoirs with the original temperatures above the cricondentherm. In addition, the field surface facilities, separators, gas plant, etc., are in the two-phase region of the system’s phase diagram. This is shown in Figure 2.17. Under these conditions, as the pressure in the reservoir is reduced due to production, the reservoir never enters the two-phase region of the gas and no hydrocarbon liquids drop out in the reservoir. That is, as pressure is reduced, the hydrocarbon fluid in the reservoir remains in its gaseous state. However, as the produced gas travels up the well and to the surface, at some point in the production system it enters the two-phase region of the phase envelope and liquid hydrocarbons develop in the well or surface facilities.

2.13: Dry Gas Reservoirs
Dry gas reservoirs are gaseous hydrocarbon systems that reside in reservoirs with the original temperatures above the cricondentherm. In addition, the surface conditions are also greater than the cricondentherm. This is shown in Figure 2.18. Under these conditions, as the pressure in the reservoir is reduced due to production, both the fluids in the reservoir and the fluids in the production system remain a single gaseous phase.

2.14: Key Learnings
- Crude oil and natural gas exist in the ground in the pore spaces of reservoir rock, typically sedimentary rock.
- Crude oil and natural gas accumulate in the ground in structures known as hydrocarbon reservoirs.
- There are five elements that are required for a hydrocarbon reservoir. The absence of just one element will preclude a geological feature from acting as a hydrocarbon reservoir. The timing of the formation of these elements is critical; all elements must be in place before the migration of oil or gas begins. These elements are:
- the source rock
- the migration path
- the cap rock
- the trap
- reservoir quality rock
- The source rock is an organic-rich sedimentary rock where catagenesis converts dead organic material, predominantly dead algae and zooplankton, into kerogen and eventually hydrocarbons.
- The migration path is a pathway where mature oil and gas migrate from the source rock to the trap. During the migration process, buoyancy is the dominant force acting on the hydrocarbons.
- The cap rock is an impermeable rock that prevents lighter oil and gas from migrating vertically away from the trap and towards the surface.
- The trap is a specific geological structure that keeps the crude oil and natural gas in place over geological time (i.e., provides four-way closure to the oil and gas). Traps can either be stratigraphic in nature, where the trapping mechanisms are dominated by the layering of sedimentary rocks, or structural in nature, where the trapping mechanisms are dominated by the folding and faulting of the rocks.
- The reservoir rock is a rock that has sufficient reservoir quality to allow for commercial crude oil and natural gas accumulations:
- sufficiently high porosity to allow for the storage of crude oil and natural gas in commercial volumes;
- sufficiently high permeability to allow for the subsurface transport of crude oil and natural gas at commercial production rates.
- The prevailing theory is that the hydrocarbons that make up crude oil and natural gas are organic in nature.
- Crude oil and natural gas are complex mixtures of four hydrocarbon types:
- alkane hydrocarbons
- naphthenic hydrocarbons
- aromatic hydrocarbons
- asphaltene hydrocarbons
- The phase behavior of the complex mixtures of hydrocarbons are determined by the composition of the mixture and are governed by Gibbs Phase Rule.
- The original pressure and temperature of a hydrocarbon reservoir at discovery are determined by the local hydrostatic gradient and thermal gradient, respectively.
- Where the original pressure and temperature of the reservoir lies in the phase envelope of the phase diagram determines the type of crude oil or natural gas system that will be produced from the reservoir.
2.15: Summary and Final Tasks
Summary
In this lesson, we learned about the origin of crude oil and natural gas and their occurrence in subsurface reservoirs. In particular, we learned that subsurface reservoirs are composed of porous and permeable rocks and that there are five requirements for a crude oil or natural gas reservoir:
- the presence of a source rock where organic material accumulates and, over time, is converted to kerogen (a precursor to crude oil and natural gas) due to the subsurface heat and pressure;
- the presence of a migration path where hydrocarbons can migrate from the source rock to the reservoir;
- the presence of a cap rock to prevent the lighter oil and gas molecules from escaping from the reservoir to the surface because of their buoyancy in water;
- the presence of a reservoir rock to allow for (A) the accumulation of oil and gas in commercial quantities and (B) the producibility of fluids at commercial rates;
- the presence of a trap to provide a mechanism to contain the oil and gas over geologic time until discovery.
In addition, we discussed the two major classifications of hydrocarbon traps:
- stratigraphic traps where the trapping mechanism is dominated by the layering of the rock;
- structural traps where the trapping mechanism is dominated by the folding, faulting, and deformation of the reservoir rock.
We also saw that crude oil and natural gas is composed of hydrocarbon molecules of organic origin. These hydrocarbon molecules are categorized into four types:
- paraffinic (or alkane) hydrocarbons which are straight chain molecules (or branched-chain molecules call isomers);
- naphthenic (or saturated cyclic) hydrocarbons which are ringed molecules where all of the bonds between carbon atoms are single bonds;
- aromatic (or unsaturated cyclic) hydrocarbons which are ringed molecules where double bonds between carbon atoms are present;
- asphaltene hydrocarbons which are complex mixtures of chain and ring molecules.
We also saw that the dominant hydrocarbon type can be determined by the Watson Characterization Factor.
Finally, we discussed the basics of the phase behavior of the hydrocarbon accumulations. We saw that this phase behavior can be visualized in a Phase Diagram and can be quantified with tools, such as the Gibbs Phase Rule, among others (like Cubic Equations of State). We also saw that the phase envelopes shown in the Phase Diagrams, the initial reservoir pressure and temperature at discovery, the pressure-temperature path of the reservoir during production, and the pressure-temperature path of the fluids going up the well and through the surface equipment results in several types of hydrocarbon reservoirs. The hydrocarbon reservoirs that we discussed were:
- undersaturated and saturated black oil, or low-shrinkage, crude oil reservoirs;
- undersaturated and saturated volatile oil, or high shrinkage, crude oil reservoirs;
- gas condensate reservoirs (sometimes referred to as retrograde condensate reservoirs) where liquid hydrocarbons condense from the gaseous phase;
- wet gas reservoirs;
- dry gas reservoirs.
Final Tasks
Reminder - Complete all of the Lesson 2 tasks!
You have reached the end of Lesson 2! Double-check the to-do list on the Lesson 2 Overview page [4] to make sure you have completed all of the activities listed there before you begin Lesson 3.



